1. Tools and techniques targeting glutamate carboxypeptidase II (GCPII)
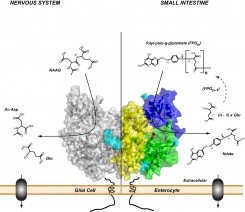
GCPII is a membrane-zinc metallopeptidase implicated in several physiological processes. Within the nervous system, GCPII exerts its peptidase activity by hydrolyzing N-acetyl-aspartyl-glutamate and the jejunal form of the enzyme acts as folate hydrolase, thus participating in the absorption of dietary folates. GCPII-specific inhibitors have been reported to be neuroprotective in multiple preclinical models of neurodegeneration. Additionally, given the GCPII over-expression in prostate carcinoma and within the neovasculature of solid tumors, the protein serves as an attractive target for imaging and therapy of a variety of cancers.
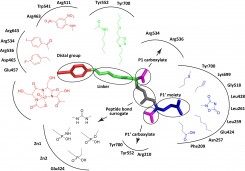
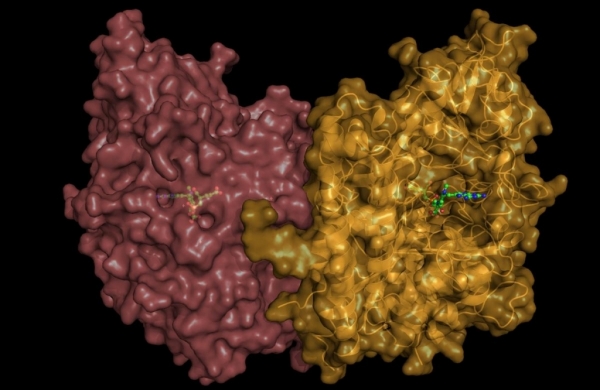
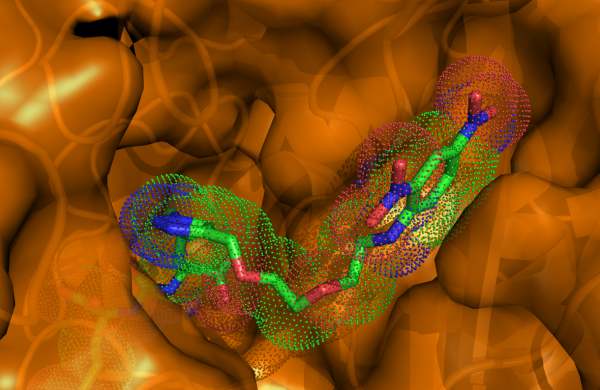
Our efforts are focused on addressing questions pertaining to the fundamental (patho)physiological roles of GCPII in healthy tissues as well as in cancer and neurodegeneration. We solved the first high-resolution X-ray structure of human GCPII and these data, together with more than 40 reported structures of GCPII-inhibitor complexes, serve as a basis for our extensive program aimed at the structure-assisted design of novel inhibitory compounds targeting GCPII.
In addition to projects involving small-molecule GCPII inhibitors we are actively pursuing the development of biologics (Anticalin-based scaffolds) as novel agents for prostate cancer imaging. In our basic-oriented research we aim at unraveling putative non-enzymatic role(s) of human GCPII (receptor, chaperone) and defining functions of GCPII orthologs in several model organisms, including yeast, Schistosoma sp, A. thaliana and C. elegans.
2. Structure-function studies of histone deacetylases (HDACs)
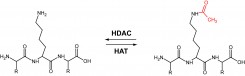
Lysine acetylation is a major post-translational modification that plays a key role in many physiological processes and is believed to have a broad spectrum of modulatory functions within the cell. Acetylation levels of cellular targets are regulated by opposite activities of histone acyltransferease and histone deacetylases (HDACs). Eighteen human HDACs identified so far can be divided into four major classes and our laboratory is specifically interested in class IIb (HDAC6, HDAC10) and class IV (HDAC11) proteins.
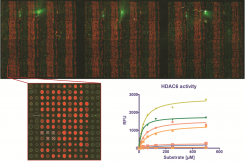
To address basic questions related to the structure-function relationship of these hydrolases we employ a panel of molecular biology, biochemistry and enzymology techniques targeting the individual HDACs as well as their cognate substrates. We have implemented several expression systems in our lab allowing us to purify milligram quantities of pure HDACs.
3. Collaborative projects
In addition to our own research, we actively participate in several collaborative projects that take an advantage of a portfolio of techniques established in our lab. These techniques include high-throughput heterologous protein expression in several systems including E. coli, K. lactis, insect S2 and Hi5 cells, and mammalian HEK293 cells, enzymatic and physicochemical characterization of purified proteins and X-ray structure determination, including a robotic screening of crystallization conditions.
Grant support: